International collaboration | foreign component in research
What is NIH’s definition of a foreign component?
The performance of any significant scientific element or segment of a project outside of the United States, either by the recipient or by a researcher employed by a foreign organization, whether or not grant funds are expended.
Activities that would meet this definition include, but are not limited to:
- The involvement of human subjects or animals
- Extensive foreign travel by recipient project staff for data collection, surveying, sampling, and similar activities, or
- Any activity of the recipient that may have an impact on U.S. foreign policy through involvement in the affairs or environment of a foreign country.
Examples of other grant-related activities that may be significant are:
- Collaborations with investigators at a foreign site are anticipated to result in co-authorship
- Use of facilities or instrumentation at a foreign site; or
- Receipt of financial support or resources from a foreign entity.
Foreign travel for consultation is not considered a foreign component.
If you are collaborating with a foreign entity to develop, perform, analyze, and disseminate research, you must indicate this information in a few places when you submit a grant application to NIH.
1. On the Application Certification for External Funding, mark the International Collaboration box towards the top of the form, along with any other Resources that pertain to the application:

2. On the NIH application itself, in the Other Project Information Form, a question asks:
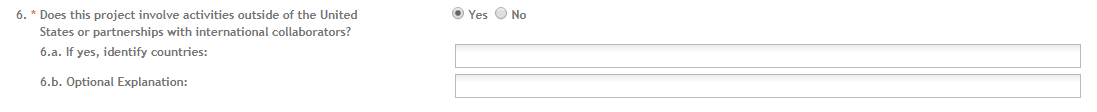
This field is required. Indicate whether this project involves activities outside of the United States or partnerships with international collaborators. Check “Yes” or “No.”
Applicants to NIH and other PHS agencies must check “Yes” if the applicant organization is a foreign institution or if the project includes a foreign component. See NIH Glossary for a definition of a foreign component.
If you have checked “Yes” to Question 6, you must include a “Foreign Justification” attachment in Field 12, Other Attachments. Describe special resources or characteristics of the research project (e.g., human subjects, animals, disease, equipment, and techniques), including the reasons why the facilities or other aspects of the proposed project are more appropriate than a domestic setting. In the body of the text, begin the section with a heading indicating “Foreign Justification” and name the file “Foreign Justification.”
If you answered “Yes” to Question 6, enter the countries with which international cooperative activities are planned. You may use abbreviations. Your entry is limited to 55 characters.
6.b. Optional Explanation: This field is optional. Enter an explanation for involvement with outside entities. Your entry is limited to 55 characters.
3. Biosketch: Foreign faculty appointments and all external support including foreign grants
On your Biosketch, you must list if you have foreign faculty appointments in the current positions section. Additionally, if you have received external funding from a foreign entity or collaborated on a foreign grant in the past three years, that information must be clearly displayed in the Research Support section of your biosketch.
4. Conflict of Interest Commitment Reporting
You must seek pre-approval to engage in an external employment, such as a dual faculty appointment, or a consulting relationship with an external entity, including all foreign institutions.This information may also be indicated on the individuals’ Annual Conflict of Interest Disclosure Form. The activity specifics are discussed in the self-written “Brief Description” which is submitted as part of the COI disclosure request packet as supporting documentation. For more information on Conflicts of Interests and Commitment visit the Compliance Office’s website HERE.
